Articles from NAMSA

NAMSA, a world-leading MedTech testing, clinical and regulatory consulting firm, announced today that it has entered into a definitive agreement to acquire the U.S. medical device testing operations of WuXi AppTec, a global company that provides a broad portfolio of R&D and manufacturing services to enable the pharmaceutical and life science industries to advance discoveries and deliver groundbreaking treatments to patients.
By NAMSA · Via Business Wire · January 17, 2025

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering end-to-end market access services, and TERUMO, a global leader in medical technology, announced that they have entered into a strategic outsourcing partnership aimed at accelerating the regulatory approval and commercialization of Terumo’s product portfolio.
By NAMSA · Via Business Wire · October 15, 2024

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today its acquisition of CRI - The Clinical Research Institute, a German-based full service CRO.
By NAMSA · Via Business Wire · July 18, 2023

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, and Terumo Aortic, a developer of aorta-focused medical devices, announced that they have entered into a strategic outsourcing partnership to assist with the acceleration and commercialization of Terumo Aortic’s innovative aortic disease products.
By NAMSA · Via Business Wire · June 21, 2023

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today its acquisition of SUAZIO, an EU-based strategic consultancy providing global market research services to medical device and IVD developers.
By NAMSA · Via Business Wire · March 27, 2023
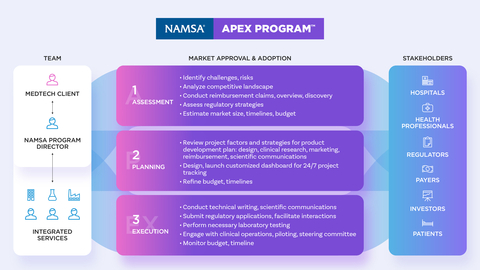
NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today the transformation of its integrated MedTech commercialization solution: the NAMSA APEX Program™. The relaunch and transformation of the Program reflects NAMSA’s continued focus on strategic outsourcing solutions to address development challenges encountered by MedTech Sponsors, resultant from current market conditions and demands.
By NAMSA · Via Business Wire · November 17, 2022

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today its acquisition of Perfectus Biomed Group, a UK-based laboratory providing customized microbiological services.
By NAMSA · Via Business Wire · October 4, 2022

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, and InspireMD, Inc. (Nasdaq: NSPR), developer of the CGuard™ Embolic Prevention System (EPS) for treatment of carotid artery disease and prevention of stroke, announced today that they have entered into a strategic outsourcing partnership to accelerate medical device development and commercialization.
By NAMSA · Via Business Wire · September 8, 2022

NAMSA, a world-leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today its nearing acquisition of Medanex Clinic, a Belgium-based European preclinical research organization.
By NAMSA · Via Business Wire · March 30, 2022

NAMSA, a world leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today its intent to acquire ÅKRN Scientific Consulting, a prominent European Medical Device CRO based in Madrid, Spain.
By NAMSA · Via Business Wire · March 21, 2022

NAMSA, the world’s leading MedTech Contract Research Organization (CRO) offering global end-to-end development services, announced today the expansion of its Minneapolis preclinical research laboratory. This expansion will add 55,000 sq. ft. to the current 130,000 sq. ft. preclinical campus in Minneapolis.
By NAMSA · Via Business Wire · November 11, 2021

NAMSA, the world’s only 100% medical device-focused Contract Research Organization (CRO) providing full continuum development solutions, announced today its acquisition of Clinlogix, a leading, Philadelphia-based global clinical research organization. Third in an advancing series, this acquisition follows NAMSA’s purchase announcements of Syntactx and American Preclinical Services (APS) in early 2021.
By NAMSA · Via Business Wire · August 2, 2021

NAMSA, the world’s only 100% medical device-focused Contract Research Organization (CRO) providing full continuum development solutions, announced today that the U.S. Food and Drug Administration (FDA) has granted the organization Accreditation Scheme for Conformity Assessment (ASCA) status. The ASCA Pilot Program, a first-of-its-kind conformity assessment program created by the U.S. FDA, reduces the regulatory burden on medical device manufacturers through consensus of biocompatibility testing requirements for efficiency.
By NAMSA · Via Business Wire · July 28, 2021
